Surface Stress
To fix the idea of surface stress, first consider an elemental crystal. The atoms at the surface of the crystal have a different bonding environment from those in the bulk. Consequently, the energy per atom at the surface differs from that in the bulk, giving rise to the surface energy. The bonding difference also causes a residual stress field in the first few monolayers at the surface, which is called surface stress. More precisely, the surface energy density is the excess energy per unit surface area. The surface stress is the variation of the surface energy density with the variation of the strain parallel to the surface, keeping the number of lattice sites constant on the surface.
For an epilayer less than a few monolayers thick, it is impossible to attribute the energy to individual kinds of misfit. Instead, we lump the whole epilayer, plus the adjacent monolayers of the substrate, into a single superficial object, and specify the energy for this object. In a way similar to elemental crystal surface, we can define surface energy and surface stress for an epilayer.
Surface stress causes phase refining. When the monolayer is a two-phase mixture, each phase has its own surface stress. The difference in the surface stresses in the two phases induces an elastic field in the substrate, and thereby changes the free energy of the system. The following figures illustrate the concept. Imagine a “cut and paste” operation. Start with a pure a phase monolayer on the substrate (a), and a pure b phase monolayer on the substrate (b). In both structures (a) and (b), the surface stresses are uniform, denoted as and and . To be definite in the following discussion, say . To be definite in the following discussion, say 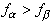 and let and let  . .
|
We then relax the structure from state (c) to state (d) by gradually releasing the applied force  . In the process, the a phase contracts, the b phase expands, and the substrate deforms accordingly. The release of the force . In the process, the a phase contracts, the b phase expands, and the substrate deforms accordingly. The release of the force  reduces the net elastic energy stored in the system. reduces the net elastic energy stored in the system.
|
Evidently, the more refined the phases, the more elastic energy can be reduced - that is, the concentration-dependent surface stress causes phase refining. On the other hand, as the phases refine, more phase boundaries are introduced into the monolayer, raising the total free energy. It is the competition between the coarsening due to the phase boundary energy and refining due to the nonuniform surface stress that selects a phase size. The quantitative analysis shows that this competition gives rise to the nanoscale phase size. Furthermore, the elastic field in the substrate leads to spatial ordering of the phases in the monolayer.
|

