

A Diver's Guide To Oxygen Therapy Apparatus
by
Larry
"Harris" Taylor, Ph.D.
This
is an electronic reprint and expansion of an article that appeared in Sources (Part
1: July/Aug. 1989, 30-35 & Part 2: Sept/Aug 1989, 72-74). This material is
copyrighted and all rights retained by the author. This article is made
available as a service to the diving community by the author and may be
distributed for any non-commercial or Not-For-Profit use.
All rights reserved.
Go To: Home About "Harris" Articles Slides War Stories Editorials Links Fini
Jump To: Cylinders Regulators Adaptors Masks Recommendations
Every
diver has been told that recompression is the treatment of choice for a serious
diving malady. However, since divers may dive in locations separated in time and
space from recompression facilities, there is often an appreciable delay (12-40
hours, or more) between the onset of the first recognizable symptoms and
admission to a recompression facility. Therefore, it is important for divers to
accept the reality that they, on the dive site, as the first responders,
determine the subsequent quality of life (if life continues) long before
professional medical people see the victim. Divers must realize that in the
field, without professional medical care, the administration of 100 % oxygen is
the treatment of choice for dealing with most serious diving maladies.
Oxygen
is often used in traumatic injuries to treat shock-associated hypoxia (lack of
oxygen in the body tissues due to a decrease in blood circulation) or to
supplement the breathing of those with chronic lung disease. The vast majority
of oxygen equipment available to the public is designed to manage those
particular problems. These devices have saved lives, and they will continue to
do so, in situations that they were designed to manage. However, devices that
were designed for home treatments are inadequate for the dive accident scenario.
In diving problems, the key to successful management of the diving accident
victim pending hyperbaric oxygen therapy is continued breathing of the highest
possible concentration of oxygen en route to professional medical
assistance.
A
serious dive malady can be described as "bubble trouble." A
large bubble(s) of gas, composed primarily of nitrogen, an inert gas, has formed
and that bubble(s) interferes with normal body functioning.
(Note: In decompression sickness a nitrogen bubble forms as the
supersaturated nitrogen gas comes out of solution; in an air embolism the bubble
initially is air (79 % nitrogen). However, the body's metabolism uses the oxygen
in the air bubble, so that eventually the bubble becomes almost all nitrogen. In
decompression sickness, the bubble can form in almost any tissue. In a lung over
pressurization event, the bubble of air is injected into the spaces around the
lung or directly into arterial circulation. If the bubble enters the arterial
circulation, the bubble generally first lodges in a capillary. Since blood flow
is only one way, cells downstream from the bubble stop receiving nutrients and
oxygen, while cell-toxic metabolic waste products begin to build-up. The result
is that cells downstream from the bubble begin to die.
Many cells (particularly those of the central nervous system), once dead,
are never regenerated. If too many
cells die, life is either impaired, or in the worst case, ceases.
Treatment
for "bubble trouble" is to reduce the size of the bubble so that the
bubble can no longer interfere with normal function or to move the bubble
through the capillary and thus restore blood flow.
This can be accomplished physically in a recompression chamber.
The effect of increased pressure (typically, 165 fsw in air embolism
cases and 60 fsw in decompression sickness) is to decrease the volume of the
offending bubble. Decreasing bubble size can also be accomplished by the
administration of a very high concentration of oxygen.
We
use oxygen primarily not to treat trauma-associated hypoxia, but rather as a
technique to "denitrogenate" the body. Ideally, we wish to initially
surround the offending nitrogen bubbles(s) with a pure oxygen environment. Since
we begin with a bubble composed almost exclusively of nitrogen, if we surround
the bubble with 100% oxygen, then nitrogen in the bubble will move out of the
bubble into the nitrogen-poor surroundings. (Remember Dalton's law of partial
pressure and that all gases wish to remain uniform in concentration through the
volume they occupy). The movement of nitrogen out of the bubble is controlled
primarily by partial pressure differences. The greater the difference of partial
pressure between the outside and inside of the bubble, the faster this movement
of gas will occur. Movement of gas will continue until the partial pressure for
the gas is the same inside as it is outside the bubble for all gas components
within the bubble. If the bubble's
surroundings can be kept at a high oxygen concentration, eventually the bubble
will have little nitrogen present. Oxygen (having a higher concentration outside
the bubble) will move into the bubble. However, The loss of nitrogen (moves out
because of partial pressure gradient between inside and outside the bubble) and
the consumption of oxygen by cellular processes cause the bubble to shrink in
size. Ultimately, the bubble shrinks enough to restore normal function or, if in
a capillary, move through the capillary and thus restore blood flow. In order
for this process to work, the oxygen concentration administered to the diving
accident victim must be exceptionally high, as close to 100% as possible. In
other words, to be effective in reducing the bubble (thus, saving body functions
or life itself), our oxygen administration equipment should be capable of
delivering 100% oxygen while meeting 100% of the patient's respiratory
requirements. (See Why
100% for a more detailed discussion)
There
is an enormous diversity of devices on the market for home utilization of
oxygen. These devices vary in the amount and percentage of oxygen that can be
effectively administered. Remember, the primary concern in the field management
of a diving problem is the ability to deliver 100% oxygen to the patient. Let's
examine the equipment available today:
Cylinders
are available in either aluminum or steel in a variety of sizes. Cylinders
should be rated by D.O.T. for oxygen service and painted green. Merely painting
a scuba tank green is not a good practice; Cylinders and all equipment rated for
oxygen service undergo a special cleaning to ensure that no grease or oily
combustible material is present in the system. The cylinder should be equipped
with a medical oxygen valve, which has two holes that mate to two pins on the
oxygen regulator. These valves are
commonly opened with a special wrench, although more expensive units will have a
convenient handle. Some valves have a cylinder pressure gauge built-in, thus
allowing the cylinder pressure to be monitored without having to put on the
regulator. For divers, the minimum size should be a 412 liter (14.6 cu. ft.)
steel D cylinder. The comparable aluminum cylinder is 415 liters. This will
provide 30 to 40 minutes of 100% oxygen delivered by demand valve. It will also
provide about 27 minutes of lower concentration oxygen when delivered to a
constant flow device at a minimum flow rate of 15 Liters/minute (L/min). The
standard kit now contains an aluminum Jumbo D (636.8 Liters; 22.4 cu. ft.)
cylinder. This provides about 50 minutes of use by demand system and
approximately 42 minutes of use at 15 Liters per minute. Also available is a
larger E cylinder that has a volume of 680 liters (24.1 cu. ft.). Larger
cylinders (3000 L) are available, but these cylinders are not very portable.
Cylinders should be filled only with medical (U.S.P.) grade oxygen. Some people
have filled oxygen bottles used in rescue training with compressed air because
it is cheaper. Not only is this
practice illegal, but also it could be dangerous, as compressed air is 79 %
nitrogen. In an emergency, someone
could get confused and administer compressed air (21 % oxygen) to a patient who
desperately needs 100 % oxygen.


Tank
wrench, standard valve & valve with handle
Typical constant flow system
The
preferred system is a demand mask system. These systems deliver oxygen only when
the patient breathes (demands a gas supply) and thus, the patient most
efficiently uses the gas in the cylinder. This efficiency, coupled with its
ability to furnish nearly 100 % oxygen to the patient is why the demand system
is the most desirable oxygen administration system available for use by divers
in treating a serious "bubble trouble" event. A demand regulator
(analogous to a scuba second stage) requires a high-pressure regulator
(analogous to a scuba first stage). There
are several of these devices available. The "first" and
"second" stages can be purchased separately or combined as part of a
demand valve system. Divers can consult their local hospital supply vendor for
descriptions and specifics of available units. (In dealing with local vendors,
the key word to remember is "demand"; some very good resuscitators on
the market do not have the demand feature.) In addition, DAN has assisted in
making available to divers a complete oxygen administration kit that includes a
demand regulator system. Information on this system is available from DAN
The
demand system is very similar to a scuba regulator. When the patient inhales,
the second stage senses the decrease in pressure and initiates rapid gas flow.
Since this system is sealed from the environment, this mask can deliver
approximately 100 % oxygen to the patient.
The demand mask system is the current preferred method of oxygen
administration to a breathing patient for the management of a serious diving
malady.


DAN
Oxygen Inhalator Valve
Typical Variable Rate/Constant Flow
VARIABLE
RATE/ CONSTANT FLOW SYSTEMS:
If a demand system is not used (they are expensive), then the next best option
would be an adjustable-flow medical oxygen regulator capable of delivering at
least 15 L/min. These regulators deliver a constant flow of oxygen at a rate
that can be adjusted by the first responder. Additionally, many demand systems
have a first stage that will only furnish gas to a second stage demand
regulator. In this case, a separate regulator capable of delivering a minimum of
15 L/min will be needed for the pocket mask to cope with a non-breathing victim.
Ideally, a regulator (like that contained within the DAN kit) should be
purchased which will supply both a demand valve mask and a constant flow system.
Most of the oxygen delivery systems discussed below will be utilized at minimum
of 15 L/min, but you want a regulator able to deliver more than the minimum.
Most regulators have two gauges: a cylinder pressure gauge and an oxygen
flow meter. Simply turning a knob can vary the gas flow.
Those regulators used with a cylinder whose valve contains a cylinder
pressure gauge may only have a flow meter.
Although these systems can, with the proper mask, deliver a reasonable
concentration of oxygen, the continuous flow wastes much of the gas supply and
is not nearly as effective or efficient as the demand valve in meeting the
respiratory requirements of the diving accident victim.
All
regulators should be rated for oxygen service and used only on oxygen cylinders.
They should be inspected and serviced annually by a qualified technician.
FIXED
RATE / CONSTANT FLOW SYSTEMS:
These units typically have a small (often disposable) gas bottles, an on-off
valve, and a constant flow delivery of only 6 L/min. These devices often come
with only a simple facemask. These units are not capable of delivering the
desired high concentrations of oxygen needed to "denitrogenate" and as
such, divers should consider them inadequate for their needs in dealing with any
diving malady. Although these devices are the least expensive oxygen
administration tools available, they are also the least desirable in terms of
effectiveness in dealing with a diving "bubble trouble" emergency.
There
are commercial and homemade devices available for utilizing scuba regulators
with medical oxygen cylinders. This defeats the primary purpose of safety
standards that have historically been promulgated to prevent accidents. Many
industrial or medical gases are chemically reactive. In addition, there are an
abundance of different gases used in medicine. Having standards, which allow
regulators to be used on cylinders of only one type of gas, prevents formation
of dangerous chemical combinations arising from the mixing of these different
gases. It also guarantees that the gas delivery system is chemically compatible
with the gas being delivered. Finally, this set of standards ensures that the
gas being delivered is what you think it should be.
Oxygen
is chemically reactive and can interact with a great many compounds. These
oxidation reactions liberate heat, enough to initiate combustion or, in some
cases, explosions. To prevent accidental chemical reactions from occurring, all
oxygen equipment must be rigorously cleaned and degreased. Most scuba diving
regulators are not kept scrupulously clean; thus, using a scuba regulator as an
oxygen delivery device poses an element of fire risk. There has been at least
one reported incident of a fire that occurred because of the use of a scuba
regulator and a homemade adaptor on an oxygen cylinder.
There
is always a group of divers looking for a way to "beat the system." In
the Great Lakes we have divers diving well beyond sport diving depths. Many of
these divers are "experimenting with their spinal cords" by using
homemade oxygen decompression procedures in the water. Some of these divers are
unaware of the risks of breathing 100% oxygen at depth. Although this population
is, admittedly, small, an adaptor does provide a potential for abuse by those
few people who wish to use oxygen for their in-water decompression procedures.
In-water use of oxygen should remain outside the realm of traditional sport
diving.
Some of these oxygen-to-scuba adaptors have proven of tremendous value in remote parts of the world. Most North Americans are not that isolated and can have ready access to proper equipment. Lastly, if you already have an oxygen cylinder, a suitable medical oxygen regulator and a demand valve are not much more in cost than the most often used adaptor.
Delivery Masks
The
delivery systems discussed below may supply higher concentrations of oxygen in a
clinical setting when used by clinical professionals, but, in the field, in an
emergency use by the lay community, it is reasonable to believe the
concentrations of oxygen delivered will be significantly less than text-book
quoted values
CONSTANT
FLOW DELIVERY DEVICES:
The mask used will determine the final concentration of constant flow oxygen
that the patient breathes. The gas coming out of the cylinder is 100 % oxygen;
the concentration of oxygen the patient actually breathes will depend on how
much the oxygen is diluted with air or the patient's exhalation. The final
concentration of oxygen delivered will be affected by the actual flow rate, the
quality of the seal of the mask around the patient's mouth and nose, and the
patient's rate and depth of breathing. In general, the higher the flow rate, the
higher the concentration of oxygen the patient will breathe (and the shorter the
time that the gas supply will last). The poorer the mask-patient seal, the lower
the concentration of oxygen that the patient will receive. In the field, the
flow rate and the quality of the mask seal are probably the most important
elements in determining the actual concentration of oxygen delivered to the
patient. Note that the concentrations of oxygen mask delivery systems listed
below are practical values obtained by skilled medical personnel in a clinical
setting. Values obtained in the field from highly stressed first responders will
probably be lower.
NASAL
CANNULA:
This device delivers an unpredictable amount of oxygen ranging from 24-32 % at 1
- 6 L/min depending on how much the patient inhales through the mouth. Higher
flow rates are uncomfortable for the patient. A high flow rate can quickly dry
out the nasal mucosa and become rapidly uncomfortable.
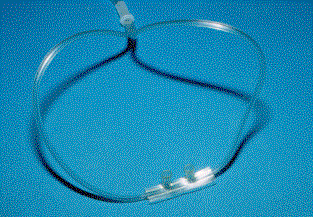
Nasal
Cannula
Simple Face Mask
SIMPLE
FACEMASK:
This device is probably the most commonly available to the public.
It seals poorly and its large ventilation holes allow the oxygen flow to
be diluted with air. The simple facemask at an oxygen flow of 6 L/min delivers
approximately 35-40 % oxygen. Increasing the flow to 10 L/min may increase
oxygen concentration to about 50 %. If the flow rate is less than 6 L/min (as
cylinder nears empty), the patient may re-breathe much of his own exhalation and
thus, the concentration of oxygen delivered will be low, possibly severely
hypoxic.
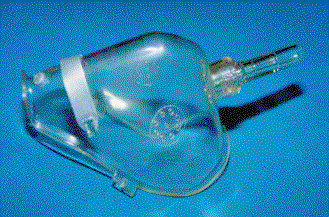
VENTURI MASKS: This device utilizes a mechanical venturi effect to increase oxygen flow rate into the mask; this limits the dilution of the oxygen by air entering into the mask. There are different types of venturi masks available. Typically, these units deliver 24 - 28 % oxygen at 4 L/min and 35 - 40 % oxygen at 8 L/min. This mask should only be used in a clinical setting and should not be used in the field.

Venturi
Mask
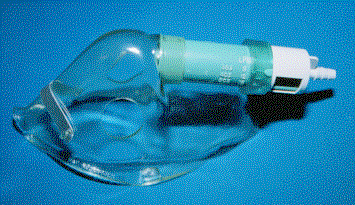
Partial Rebreather or Medium Concentration Mask
PARTIAL REBREATHER: This mask adds a reservoir bag to the simple facemask. This mask appears similar to a non-rebreather mask. However, it is missing a one-way valve between the reservoir bag and the mask. The reservoir bag fills with oxygen. When the patient breathes, much of the inhalation volume comes from the bag and thus, the oxygen concentration delivered is increased. However, with this system the patient's exhalation can mix with gas in the bag. At 6 L/min this system delivers 40-50 % oxygen; at 10-15L/min the partial rebreather can deliver approximately 60 % oxygen. These masks are often called medium concentration oxygen delivery masks.
NON-REBREATHER:
This mask consists of a mask that has a reservoir bag attached. The bag is
separated from the mask by a one-way valve that prevents air and patient
exhalation from diluting the oxygen in the reservoir bag. When the patient
inhales, the valve opens and the patient breathes primarily oxygen. There are
also one-way valves that cover the holes on the mask to allow patient exhalation
to escape without allowing large quantities of air to enter the mask. Some masks
have this one-way valve on both sides of the mask. These masks are prescription
only. If both sides are covered and
gas flow ceases, then the patient will not be able to breathe because the valves
keep air from entering during inhalation. The common high oxygen concentration
mask has a one -way valve on only one side so that if gas flow ceases, the
patient can still breathe. At a minimum oxygen flow of 15 L/min, as long as the
reservoir bag is kept filled and a good seal is maintained, this mask can
deliver 60 - 75% oxygen to the patient
The systems described above are designed for use with a patient that is breathing. This accounts for roughly 90 - 95 % of all diving accidents. Since these devices require an inhalation effort from the patient to move the oxygen into the lungs, they will be ineffective in dealing with a non-breathing patient who is in probably the most desperate need of high concentrations of oxygen. The following devices are used for the non-breathing patient:
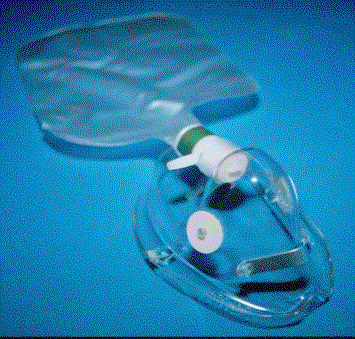
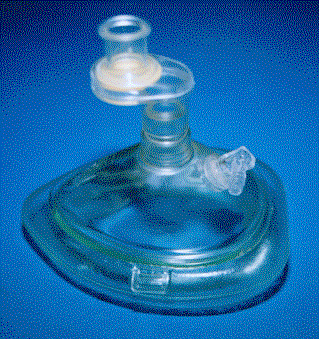
Non-Rebreather
Mask
Pocket Mask w/Oxygen Inlet & One-way Valve
POCKET
MASK:
This device is the current suggested means of ventilating a non-breathing scuba
diving accident victim. The oxygen line is hooked into a nipple on the mask, gas
flow is started, and the rescuer performs mouth-to-pocket mask resuscitation. At
6 L/min the oxygen delivered will be about 35 %. At 15L/min this procedure, with
a good mask seal, will deliver about 50 % oxygen to the patient. These units are
used in conjunction with a small lone-way valve assembly that diverts the
patient's exhalation away from the rescuer. The valve also contains a filter to
minimize risk of disease transmission between rescuer and patient. The valve
assembly should be considered single-use.
SINGLE-USE
POCKET MASKS:
This disposable device is designed for use in C.P.R. The unit (InterTech #
008010, or equivalent) comes with a single use pocket mask that forms a good
seal, a one-way valve to isolate the patient's breath from the rescuer, a short
piece of respiratory tubing, and a mouthpiece to make breathing into the device
a little easier and more comfortable. By adding a T-adaptor oxygen inlet (Airlife
U/Adapit # 004081, or equivalent) and changing the length of respiratory tubing
to longer than 12 " (the tubing acts as an oxygen reservoir; the longer the
tubing, the higher the final concentration of oxygen that can be delivered),
this new device can furnish more than 60 % oxygen to the patient at 15 L/min.
Consult your local hospital supply vendor for parts and check your assembly with
someone knowledgeable in oxygen administration to ensure that the device is
properly assembled. (See High
Concentration Delivery Mask for discussion and image)
MECHANICAL
VENTILATOR:
Many demand oxygen systems, as well as special mechanical resuscitators, can
utilize oxygen pressure to force oxygen into the lungs. Some of these,
particularly older models, do not have overpressure release valves. If too much
gas is forced into the lungs, it is possible for the patient to suffer lung
damage from the resuscitation effort. All mechanical ventilators require
specialized training and therefore belong to the realm of the licensed medical
professional. Sport divers, without special training should not utilize these
devices. Remember, a primary concern of a first responder is to do no additional
harm.
BAG VALVE MASK: This device stores oxygen in a bag that fills from an oxygen reservoir. The oxygen is delivered to the patient by squeezing the large bag. Properly used (which requires three or four hands and/or a lot of properly-supervised practice), this device can furnish nearly 100 % oxygen to the non-breathing patient. To be effective, they must be used in conjunction with an endotracheal airway and should not be used by rescuers who have not had proper training and practice to obtain this necessary skill. (Placement of an oral airway is most definitely a technique that requires training and clinical ability. Improper placement can injure or block the patient's airway. Those without clinical training and proficiency should not attempt Insertion of airway management devices.)


Bag
Valve Mask
Set of Endotrachael Airways
NASAL
CANNULA ON RESCUER:
One technique for administration of oxygen to an unconscious diver has the
rescuer breathing oxygen delivered by a nasal cannula. After breathing oxygen,
the rescuer then performs mouth-to-mouth resuscitation on the victim. Note that
this technique certainly delivers a greater concentration of oxygen to the
victim than mere mouth-to-mouth breathing. Since the nasal cannula can deliver
at best about 32% oxygen to the rescuer, the actual oxygen concentration
delivered to the patient will be probably between 20 - 30%. The effectiveness of
this technique has not yet been demonstrated.
RECOMMENDATIONS
FOR A DIVING ACCIDENT MANAGEMENT OXYGEN KIT:
1.
Avoid all 6 L/min constant flow devices.
2.
The DAN oxygen unit was assembled based on the advice of the hyperbaric medical
experts at DAN It contains a single 415 L cylinder (41 minutes supply at 10
L/min. (DAN kits now primarily use an aluminum Jumbo D (636.8 Liters;
approximately 50 minutes of gas supply for a demand inhalator)), a demand
valve delivery oxygen system for breathing patients and a pocket mask for
non-breathing patients. This kit represents the current state of the art for
dealing with oxygen administration in a serious diving emergency. As such, the
DAN kit (or its equivalent) represents the minimal amount of oxygen equipment
that should be present on dive training sites. Dive training should NOT, in my
opinion, occur with anything less than this equipment on site. Since many divers
dive or train in remote locations and are more than 30 minutes from medical
assistance, a second (or more) 415 L (or larger) cylinder is desirable. Divers
should have enough gas supply on hand to treat an injured diver for the amount
of time it takes for the local site professional emergency response team to
travel to the dive site.

Author’s
DAN Kit (with second demand system)
INSTRUCTORS
PLEASE NOTE: In the US, the DAN oxygen kit is considered the
"standard-of-care" in the diving community for treatment of diving
accidents. To train with less than this is a liability risk that you should be
unwilling to assume!
3.
A reasonable emergency alternative to the demand system, would be one (or more)
D or E cylinder, a variable flow (15 L/min) medical oxygen regulator, a high
concentration (non-rebreathing) oxygen delivery mask for breathing patients, and
either the Laerdal or single-use modification pocket mask described above for a
non-breathing patient. I tell my students to carry a non-rebreather mask in
their traveling first aid kit. So, if they are in a situation where the only
oxygen supply is constant flow and a simple facemask, substitution of the non-rebreather
mask for the simple facemask can immediately boost the oxygen concentration
given to the patient.
4.
For those who already have oxygen cylinders and possibly some oxygen
administration equipment, possible additions include:
a. A demand valve and mask with appropriate first
stage. Consult your local hospital supply vendor or DAN for advice. Many of
these devices contain a lever or trigger for positive pressure mechanical
ventilation. If your demand system
has a trigger, do not use this trigger unless you have received training in its
use. Improper use (especially with older mechanical ventilators) could damage
patient's lungs and lead to increased patient injury or death.
b. For those who already have an oxygen regulator
capable of delivering 15 L/min and cannot afford a demand mask, a non-rebreathing
mask (Hudson # 1059, or equivalent) is a necessary alternative. This mask
properly used, will deliver about 60 - 70 % oxygen at 15 L/min. The mask sells
for between $ 3 - 6. But remember, for "denitrogenation" we need
the highest possible concentration of oxygen, so, although this system is better
than nothing, all efforts should be made to supply accident management kit with
a demand system.
c. One of the pocket mask assemblies described above.
d. A rugged carrying case to protect the oxygen unit from rough handling
and the environment. Cases with O-rings to ensure watertight integrity are
recommended for use in marine environments.
Local
hospital supply vendors or DAN can be exceptionally helpful to you when you
assemble the oxygen administration portion of your emergency response kit. Make
certain that you discuss your needs with a respiratory therapist, not just a
salesperson. Explain that your oxygen use will be emergency field management of
a scuba diving injury, and that your desire is to furnish the highest possible
concentration of oxygen with only a limited supply of gas to a scuba diving
accident victim. They may also inform you about any local legal restrictions on
oxygen equipment or utilization. Note that oxygen used in aircraft, in very
small cylinders (like those sold by mail-order), or by rescue personnel for
emergency use only are exempt from prescription requirements. (Many local
vendors, however, will be more comfortable if you obtain a prescription for your
oxygen unit. My personal
prescription states that the oxygen unit is only for emergency applications in a
diving accident.)
Regardless
of what you decide to carry in your emergency response kit, remember that people
save lives; equipment merely helps. (In an emergency, divers should utilize
whatever is available to the best of their abilities within the limitations of
their training and the tools assembled.) All the above equipment is useless;
unless you (and your buddy) know how to use the life saving tools you will carry
with you to the dive site. Thus, you should seek out the training of a
knowledgeable dive rescue or oxygen administration instructor who can teach you
the proper use of oxygen administration devices. Oxygen administration is a
skill that can be easily learned, under proper supervision. Practice now could
prevent a later problem from becoming a catastrophe.
Jump To: Cylinders Regulators Adaptors Masks Recommendations
Go To: Home About "Harris" Articles Slides War Stories Editorials Links Fini
Credit:
Photography of oxygen equipment by the author using his personal arsenal of oxygen delivery equipment
About
The Author:
Larry
"Harris" Taylor, Ph.D. is a biochemist and Dive Safety Coordinator at
the University of Michigan. He has authored more than 200 scuba related
articles. His personal dive library (See Alert Diver, Mar/Apr, 1997, p. 54) is
considered one of the best recreational sources of information in North America.
All rights reserved.
Use of these articles for personal or organizational profit is specifically denied.
These articles may be used for not-for-profit diving education