| Back to Main Page | Professor Vincent L. Pecoraro | Group Information | Publications | Lectures and Conferences |
| Manganese Project | Vanadium Project | Metallacrown Project | Gentamicin Project |
| Inorganic Chemistry | Related Links |



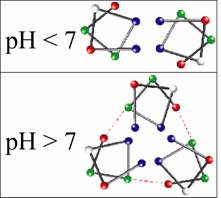 This design leads to a pH dependent structure.
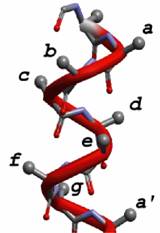
This design leads to a pH dependent structure. 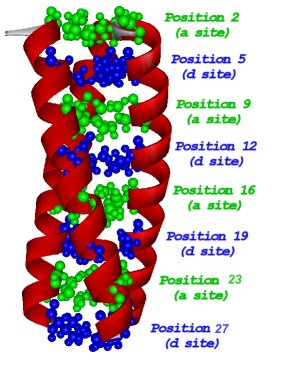 The hydrophobic residues are arranged in layers. Substitution of
Leucine
residues in a layer usually with cysteine leads to a soft metal binding
site.
The hydrophobic residues are arranged in layers. Substitution of
Leucine
residues in a layer usually with cysteine leads to a soft metal binding
site.
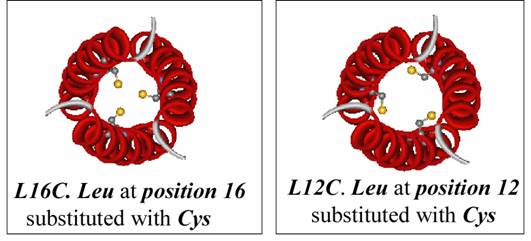
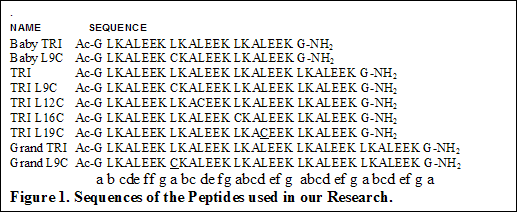
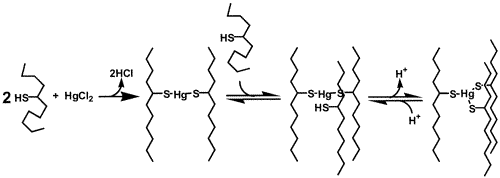
In all of the studies, the chosen metals do not bind to TRI and only
form stable complexes with cysteine substituted peptides.
Besides TRI, shorter peptides like Baby TRI, Baby L9C, Mini Tri, Mini L9C
having 4,4,3 and 3 heptads have also been synthesized and used in our
research.
Similarly longer peptide like Grand TRI and Grand L9C having one heptad
more than TRI have been synthesized and characterized.
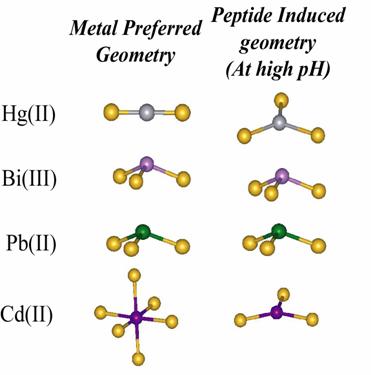
In many cases the geometrical preference of the peptide and the metal
are different.
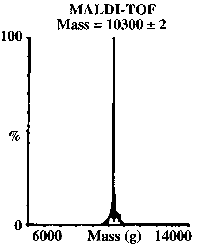

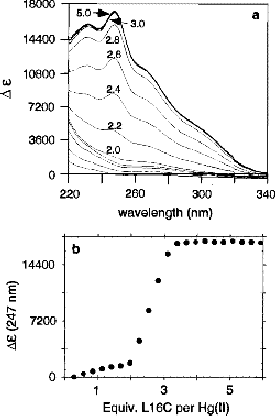
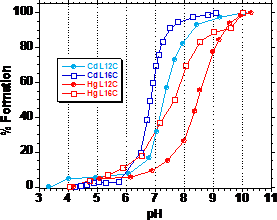
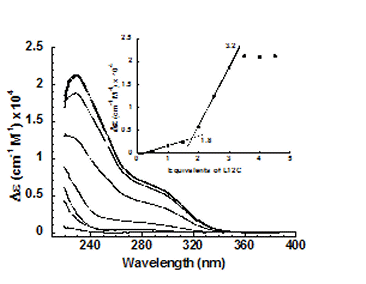
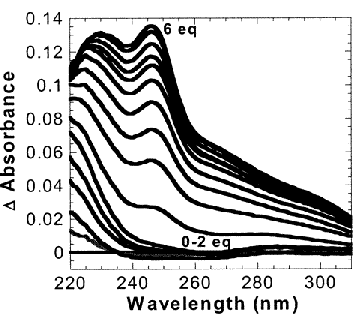
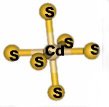
Cd(II) allowed us to assess whether the TRI peptides were capable of
lowering the preferred coordination number of a metal. Tetrahedral and
higher coordination environments are usually observed with Cd(II) in
thiolate rich environment.
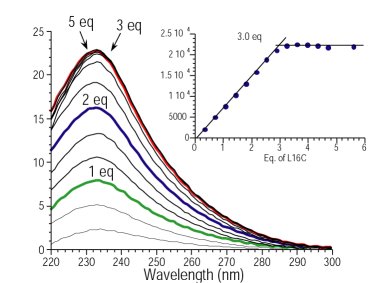



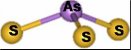
Farrer, B.T.; McClure, C.; Penner-Hahn, J.E.; Pecoraro, V.L. "Arsenic(III)-Thiol Interactions Stabilize Three-Helix Bundles in Aqueous Solution"; Inorg. Chem., 2000, 39, 5422-5423.

| Back to Main Page | Professor Vincent L. Pecoraro | Group Information | Publications | Lectures and Conferences |
| Manganese Project | Vanadium Project | Metallacrown Project | Gentamicin Project |
| Inorganic Chemistry | Related Links |