
| Back to Main Page | Professor Vincent L. Pecoraro | Group Information | Publications | Lectures and Conferences |
| Metallopeptide Project | Manganese Project | Vanadium Project | Gentamicin Project |
| Inorganic Chemistry | Related Links |


Background
Metallacrowns are inorganic analogues of crown ethers. A typical crown
ether is a cyclic oligomer with a -C-C-O- repeat unit forming the
macrocyclic ring.
In a metallacrown, a transition metal ion substitutes for one of the
carbons in
the ethylene bridge of the ether ring and a nitrogen substitues for the
other.
The repeat unit in a metallacrown, thus, becomes -M-N-O-. As shown in fig
1,
metallacrowns have similar backbone structure and ring sizes as their
organic
counterparts. To date, three sizes have been isolated:
9-MC-3,
12-MC-4, 15-MC-5, where MC
stands for
Metallacrown as an analogy to the naming system developed for crown
ethers.
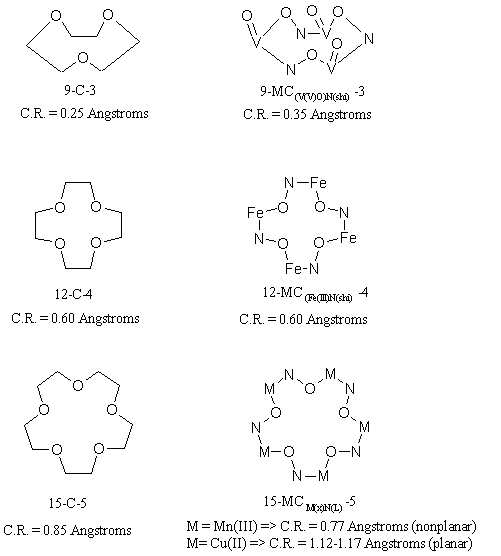
The first metallacrowns was isolated while exploring the biological
chemistry of the
sallicylhydroximate complexes of vanadium. This 1989 achievement was soon
followed
by the isolation of many complexes involving many metals,
hydroximate ligands and
ring sizes. Transition metals were used as ring metals as well as cavity
metals,
and lanthanides and actinides, now, are encapsulated by the 15-MC-5
structural
motif. In the design of the metallacrown, many considerations were taken.
The ring
size is greatly dependent on the size of ligand used and its size. The
oxidation state
of the ring metal and the number of negatively charged groups on the
ligand play a role
in limiting the number of ligands or transition metals used in synthesis.
The stability
of a metallacrown in different solvent systems is also a consideration in
the design.
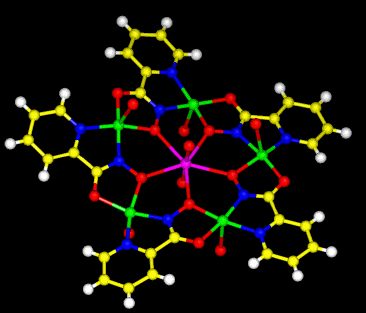
Figure 2. Crown ethers are classic ligands (composed of a cyclic polyether structure) that selectively sequester monovalent alkali metals and divalent alkali earths. The metallacrown shown here is an inorganic analogue of the crown ethers. The 15-metallacrown-5 contains Cu(II)-N-O repeats in order to form a 15-membered ring that can efficiently complex uranyl ion.

Nomenclature
Nomenclature designed for metallacrowns stem from the naming system of the
organic crown ethers.The general naming formula
is:CMn+(A-)n[X-MCM(X)T(L)-Y]
where CMn+ stand for cavity metal with its
charge shown on the upper right, and A- is the anion part of
the
cation/anion pair in the cavity of the crown. X indicates the size of the
backbone ring, and Y is the number of oxygen atoms in the ring. MC stands
for metallacrown, and L is the organic hydroxamic ligand used in
synthesis, which is usually abbreviated into the conventional two or three
letter system (for example:Picoline hydroxamic acid is abbreviated as Pic,
glycine hydroxamic acid is abbreviated as Gly...). T is a symbol for the
heterogeneous atom which is, so far nitrogen. M(X) is the ring metal with
its oxidation number shown in brackets. For example, a planar metallacrown
that resembles the 15-C-5 structural motif synthesized with Leucine
Hydroxamic acid as the ligand, copper as a ring metal, and Europium as its
cavity metal is represented as
Eu(III)(NO3-)3[15-MC-Cu(II)N(Leu)-5]
.

Project Goals

Applications and Current Work
Metallacrowns of all sizes can selectively encapsulate metals and
anions in their
cavities and are, therefore a new class of molecular recognition agents.
Those recognize not just a cation or an anion, but can also selectively,
bind cation/anion pairs. In fact, it has been shown that the size of the
metallacrown,
the solvent environment and salt bridges connecting the ring metals
with the cavity metal can effectively screen for the metal cations. for
example,
NMR studies of 15-MC-5's selectivity demonstrated the preference of those
rings for
captivating uranil ions over others, and are therefore, good candidates
for applications in the fields of identification and removal of chemical
deposits
of the cold war. The special design of metallacrowns with specific
targetting abilities
is an important goal in our lab. This involves varying the cavity size,
chirality and shape; considering different salt bridges, and studying
solvent environment under which specific results are achieved.
Metallacrowns are gaining more attention in inorganic circles. Since
cavity metals
with different properties can be selectively encapsulated by those rings,
many
applications have been proposed for metallacrowns. Trivalent lanthanide
complexes,
for example, have been shown to effectively hydrolyze phosphate diesters
and RNA.
Because the 15-MC-5 structural type encapsulates lanthanides, current work
in our
lab is exploring this new function.
Trivalent gadolinium is one of the lanthanides encapsulated by the
metallacrowns.
This cation is the most paramagentic ion known and is, therefore the best
known metal
used in MRI(Magentic Resonance Imaging) contrasting agents. Therefore, a
new
project was launched where this property of the gadolinium metallacrown is
studied.
It has been shown that metallacrowns have similar relaxivities to those
exhibited by the
MRI compounds presently in use. As a step forward in the direction of
those
biological and medical applications of metallacrowns, current work
focusses
on the stability of the 15-metallacrowns-5 in aqueous and buffered
solutions at
different pH's. Competition experiments with other popular complexing
agents, such as EDTA, and physiologically relevant molecules, such as
plasma proteins that transport
transition metals in the biological systems are being investigated.
The design of rings that are most stable under physiological conditions
includes the synthesis of metallacrowns with inert ring metals and the
study of the solution integrity exhibited by these metallacrowns.
Other applications have been proposed for the new compounds. For example,
planar
12-MC-4's are similar in size and shape to porphyrins and phthalocyanines,
which have been explored as liquid crystalline materials with diverse
applications
including data storage and directional conductivity. Metallacrowns,
therefore, as
liquid crystals is another possible destiny for the new class of compounds
as studied by the graduate students in the lab. Also the encapsulation of
specific
metals, such as cerium may amplify their catalyzing properties.
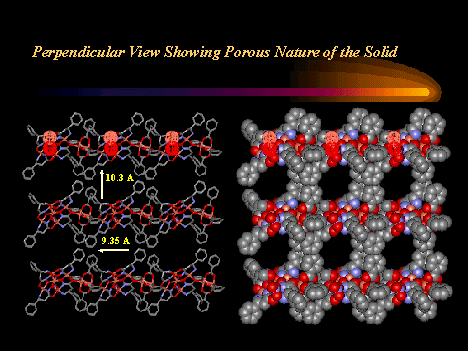
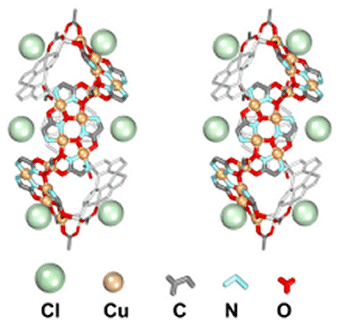
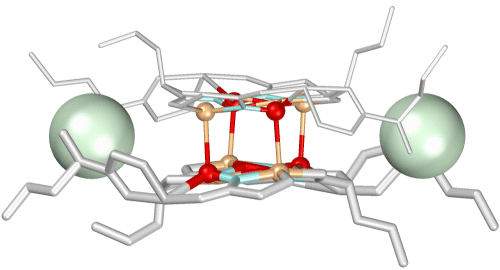
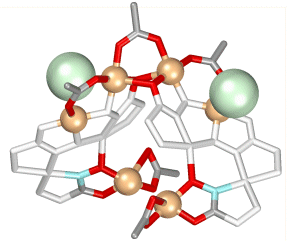
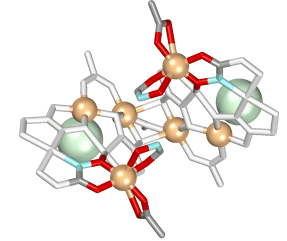
Chiral metallacrowns have been fused into two-dimensional layers using
neutral copper
benzoate paddlewheel dimers and nitrate anions to link each 12-MC--4
molecule.
(Click here for descriptive models). The solid
contains approximately 9A
channels
that are filled with solvent and a benzoic acid guest. A related chain
structure that is
generated by connecting metallacrowns with the copper dimer, but which
does not have nitrate
bridges in the second dimension, also has channels of similar dimension.
These
observations suggest a generizable strategy for the preparation of
mesoporous
chiral solids in bulk quantities.
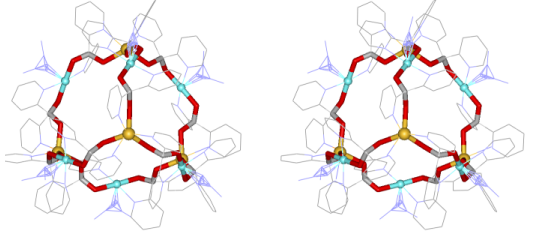
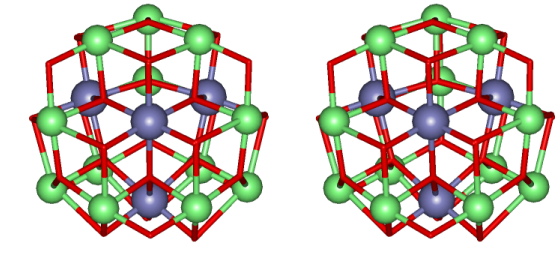
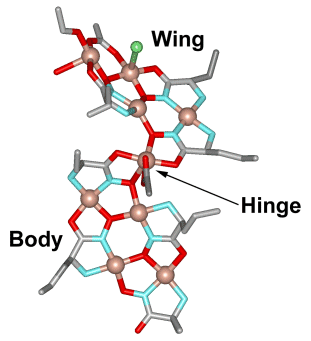
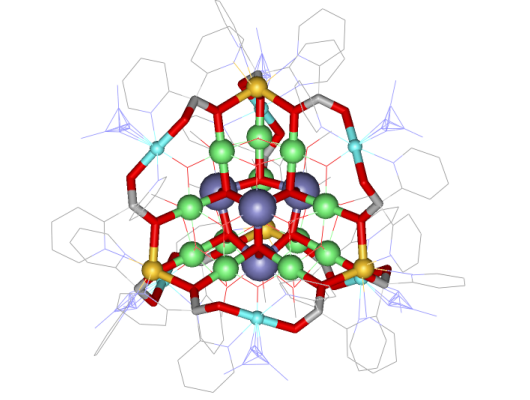 The metallacryptate
[Mn4IIMn22III(pdol)12
(µ3-OCH3)12(µ3-O)10
(µ4-O)
6(N3)6]
(pdol2-=dipyridylketonediolate ion) was synthesized by our
collaborator
Dr.
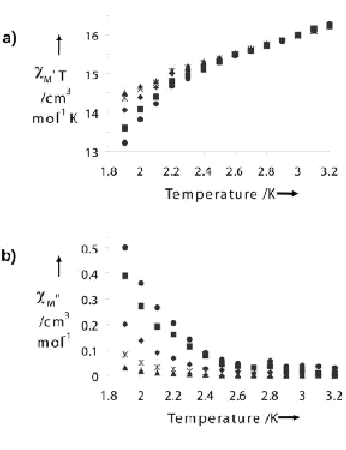
Dimitris Kessissoglou and his students in Greece. We determined the
crystal structure and its magnetic properties here in the Pecoraro group
(Angew. Chem. Int. Ed. 2003, 42, 3763).
The metallacryptate is basically
a three-dimensional metallacrown that has an encapsulated manganese
oxide
core. A strand of ten Mn centers and 12 pdol2- ligands compose
the outer
strands and the core composition is
{Mn16(O2-)12(MeO-)16}.
The metallacryptate
[Mn4IIMn22III(pdol)12
(µ3-OCH3)12(µ3-O)10
(µ4-O)
6(N3)6]
(pdol2-=dipyridylketonediolate ion) was synthesized by our
collaborator
Dr.
Dimitris Kessissoglou and his students in Greece. We determined the
crystal structure and its magnetic properties here in the Pecoraro group
(Angew. Chem. Int. Ed. 2003, 42, 3763).
The metallacryptate is basically
a three-dimensional metallacrown that has an encapsulated manganese
oxide
core. A strand of ten Mn centers and 12 pdol2- ligands compose
the outer
strands and the core composition is
{Mn16(O2-)12(MeO-)16}.

Advantages Offered by Metallacrowns over other organic and Inorganic
Ligands
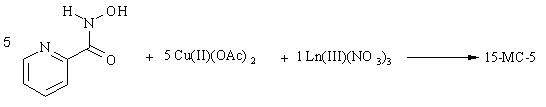

Future of the Field
Our research on metallacrowns has produced a great amount of information
about this new class of molecular recognition agents. As mentioned above,
current projects investigate the many applications proposed for the
inorganic crowns. Stability in a variety of solvents, including water,
and in buffered systems is being examined. Their solution dynamics in the
presence of other
chelating agents and biological metal sequestering proteins is being
studied.
Metallacrowns are becoming part of the inorganic and bioinorganic
communities
as new metallacrown projects are started every year at different
institutions
in the country as well as around the world (in Greece, Germany, Finland
and Korea).
The new field has a very promising future and has many inorganic and
material
as well as biochemical and biological applications. Metallacrowns are
indeed
the wave of the future!


| Back to Main Page | Professor Vincent L. Pecoraro | Group Information | Publications | Lectures and Conferences |
| Metallopeptide Project | Vanadium Project | Manganese Project | Gentamicin Project |
| Inorganic Chemistry | Related Links |