KubarychGroup
Solvation Dynamics
When a reaction occurs, the solvent responds, and simultaneously plays an active role in determining the dynamics and fate of the chemical event. Traditional methods study only the reacting species. We have studied the first solvation shell directly for the first time!
Frequency Shift Encodes Alignment
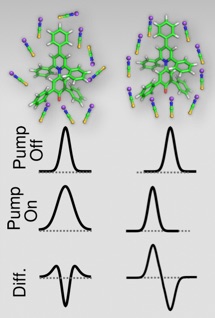
The figure to the left shows two hypothetical solvation environments that are distinguishable using dynamic vibrational Stark effect spectroscopy. In the left case, the solvent shell molecules are essentially unaligned with the main axis of betaine-30, which is the axis along which the electric field changes the most following photoinduced charge transfer. Depending on the position and alignment of the solvent molecules, they will shift either up or down in frequency more or less randomly. The random shift is illustrated by the broader (“Pump On”) spectrum. The difference, Pump On–Pump Off gives the last spectrum, showing a characteristic negative signal in the middle with two positive bands on either side.
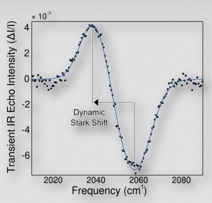
In the case where the solvent molecules are partially aligned, we see a clear difference. Here the solvent molecules are more likely to respond similarly, and rather than just broadening, the band actually shifts in frequency. So the difference shows a negative band corresponding to the ground state, with an induced positive band to lower frequency (to the left). This latter case is what we observe in our experiment (bottom figure).
It is possible to use the laser polarizations to obtain independent evidence that the molecules are aligned by comparing the signal with the visible and IR polarizations parallel to the case where they are perpendicular. We find the signal to decrease by 40% in the perpendicular arrangement relative to all-parallel, which is consistent with the Stark effect picture.
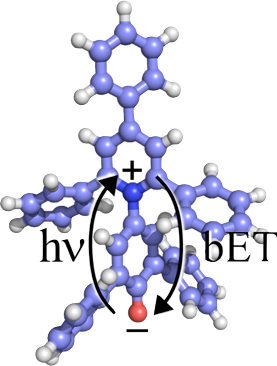
Dynamic Stark Effect
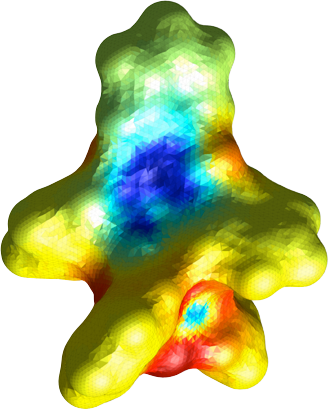
When an anharmonic oscillator is in the presence of an electric field, the transition frequency of the oscillator will change. The precise magnitude and direction of the shift depends on the electronic structure of the molecule, and can be calculated using quantum chemical methods. The lowest-order interaction leads to a linear Stark shift, and that is actually what is shown in the color scale on the surface in the image above. Using the changes in the partial charges of the atoms upon optical excitation and charge-transfer, we can calculate the change in the electric field (using Coulomb’s law) at a surface that describes the likely position of the first solvation shell molecules. Since the shift depends on orientation (that we do not know) we show the maximum magnitude of the Stark shift. The scale ranges from red (-65 cm-1) to blue (+85 cm-1).

