Research Projects
Wearable nanoelectronic transdermal sensors
Probing molecule-nanomaterial interaction
Other 2D materials
Graphene heterodyne vapor sensors
Carbon nanotube high-frequency biosensors
Wearable nanoelectronic transdermal sensors
Many diseases and human physiological conditions need to be monitored continuously for timely clinical intervention and personalized treatment. Conventional blood-based detection is invasive and prone to infection, and thus cannot be used for continuous monitoring. Many vapors (such as NH3, H2S, NO, and ethanol) from exhaled breath or transpiring from the skin reflect the conditions of various diseases (such as diabetes, peripheral artery disease, etc.). Nanoelectronic materials provide fast, sensitive, and body-pliable sensing platforms ideal to implement wearable vapor sensors for completely non-invasive and continuous health monitoring. Currently, I am working on the research and development of highly integrated wearable graphene sensors to monitor clinically relevant transdermal vapors. This work is funded by NSF SBIR Phase I grant and being carried out at my company Arborsense, Inc. and University of Michigan.
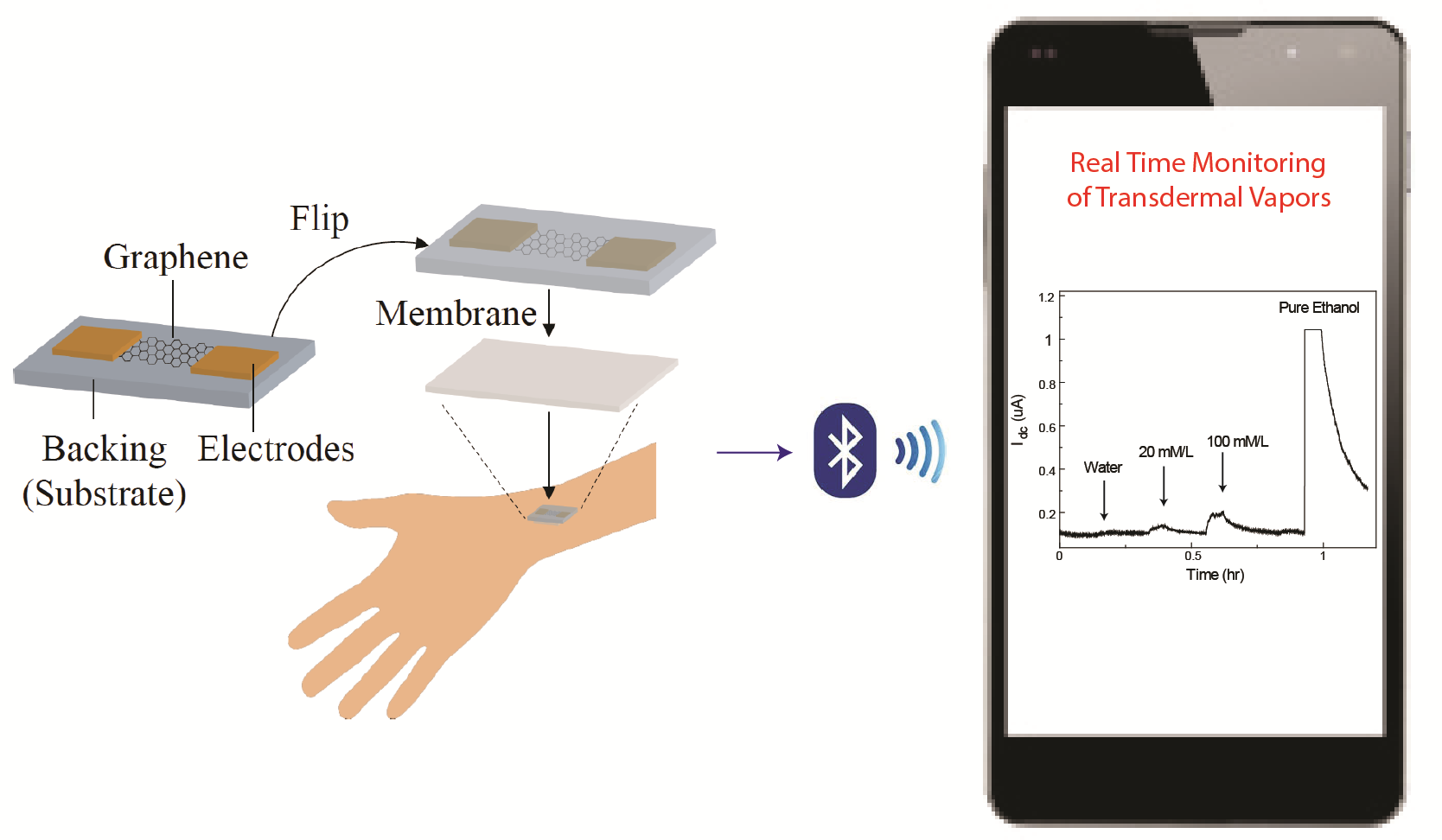
Wearable graphene nanoelectronic sensor for continuous real-time detection of clinically relevant transdermal vapors where sensor data is wirelessly transmitted to phone for personalized healthcare.
Probing molecule-nanomaterial interaction
One of the research directions I am currently pursuing is to understand the non-covalent chemical interactions between small molecules and a pristine nanomaterial surface. Non-covalent interactions, though weak (~100 meVs), are the bedrock of key chemical, biological, and cellular processes. The heterodyne sensor technology I have developed in my dissertation work provides key advantages to study such interactions. Firstly, heterodyne signal does not record charge transfer through covalent bonds; instead, it reads the dipole interactions which are the basis of weak van der Waal forces. Secondly, the fast, sensitive and reversible heterodyne responses allow us to monitor vapor dynamics near a nanomaterial surface in real-time and quantify the thermodynamic molecule-nanomaterial binding affinity using temperature-dependent desorption spectroscopy. Further, the ability to electrically tune the charge density (hence the chemical potential) in nanomaterials via an electrostatic gate can enhance or weaken the electronic interaction between a molecule and the nanomaterial, thereby providing another tool to control molecular physisorption. This ability to quantify, control, and tune non-covalent interactions can lead to rational designs of complex chemical processes for catalysis, drug-discovery, and chemical and biological sensor development.
Kulkarni et al. Nano Letters, 16 (1), pp. 695-700, 2016.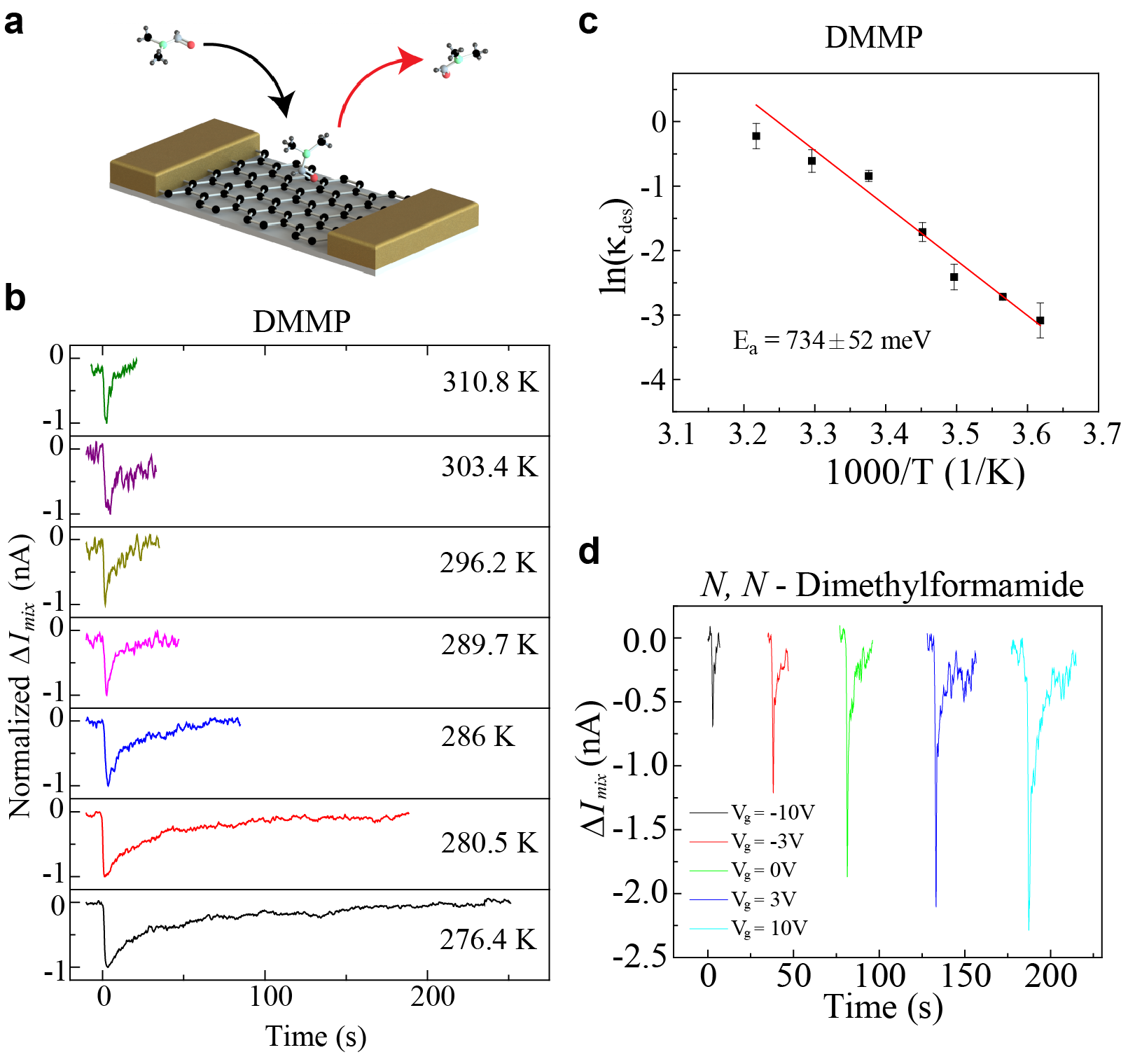
Other 2D materials
Transition metal dichalcogenides (TDMCs) offer properties that are complementary to yet distinct from those in graphene. Pristine graphene lacks a bandgap and hence, graphene field effect transistors (FETs) cannot be effectively switched off and have low on/off switching ratios. In contrast, several 2D TMDCs such as MoS2, MoSe2, WS2 and WSe2 possess sizable bandgaps around 1-2eV, that change from indirect to direct in single layers, allowing applications in nanoelectronics and optoelectronics. Substantial effort has been dedicated to understand the intrinsic electronic properties of molybdenum disulfide (MoS2). However, electron transport study on monolayer MoS2 has been challenging to date, especially at low temperatures due to large metal/semiconductor junction barriers. We have demonstrated high performance MoS2 devices through the use of low work function metal (zinc) contacts. Further,Coulomb blockade is observed at low temperatures due to single-electron tunneling via two tunnel junction barriers. These results hold promise for the study of novel quantum transport phenomena in 2D semiconducting atomic layer crystals.
Lee, Kulkarni, and Zhong. Nanoscale, doi: 10.1039/C5NR08954A.(2016).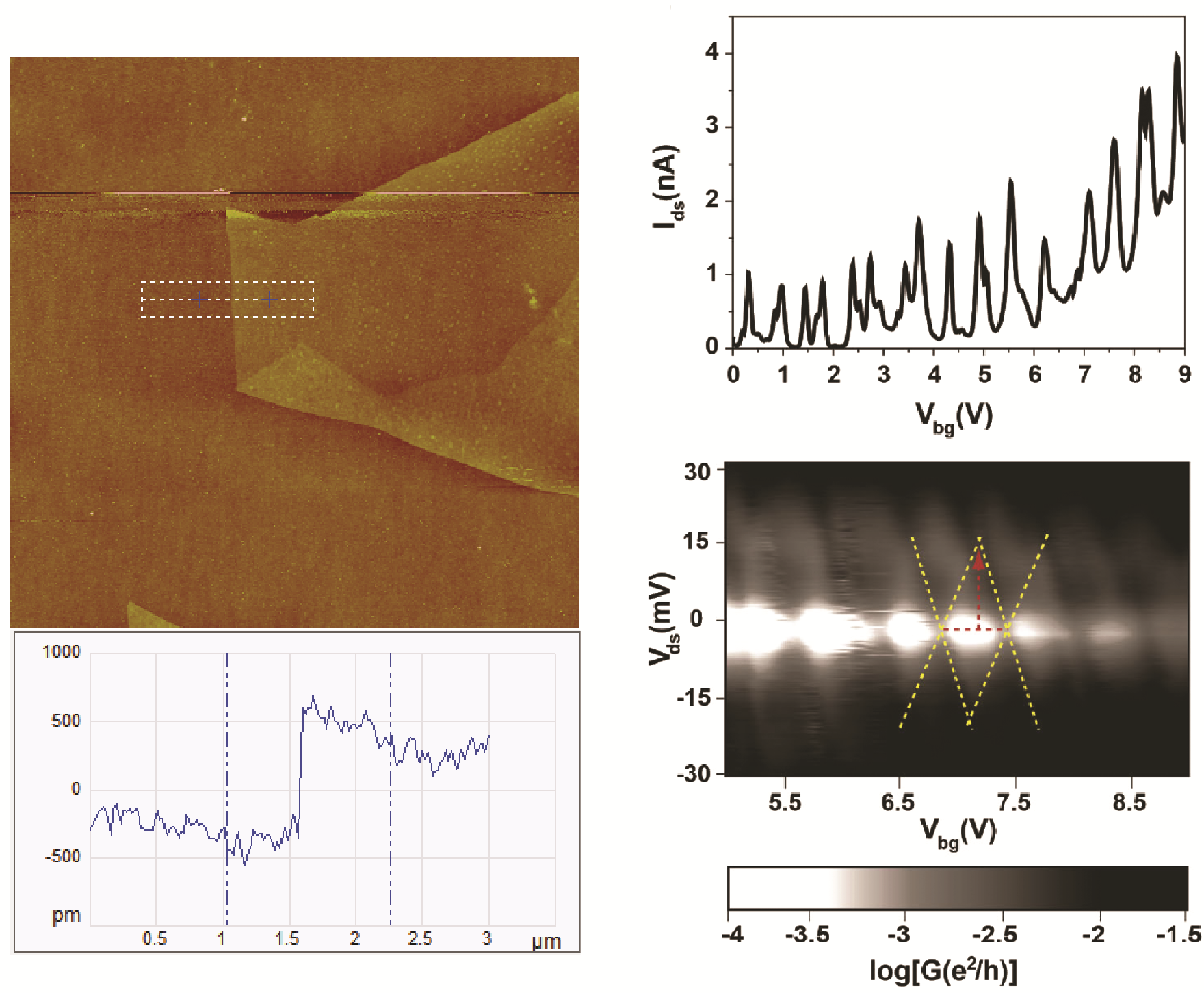
Graphene heterodyne vapor sensors
Nearly all existing nanoelectronic sensors are based on charge detection, where molecular binding changes the charge density of the sensor and leads to sensing signal. However, intrinsically slow dynamics of interface-trapped charges and defect-mediated charge-transfer processes significantly limit those sensors' response to 10s to 1000s of seconds, which has long been known as a bottleneck for many applications requiring rapid and sensitive response. We have demonstrated a fundamentally new sensing mechanism based on heterodyne detection and achieved rapid (~0.1s) and sensitive (~1ppb) detection of wide range of vapor molecules. We show that heterodyne signal is dependent on the dipole moment of the molecule rather than associated charge and hence overcomes the fundamental challenge of speed-sensitivity tradeoff faced by conventional DC detection techniques.
Kulkarni et al. Nature Communications, 5:4376 doi: 10.1038/ncomms5376 (2014).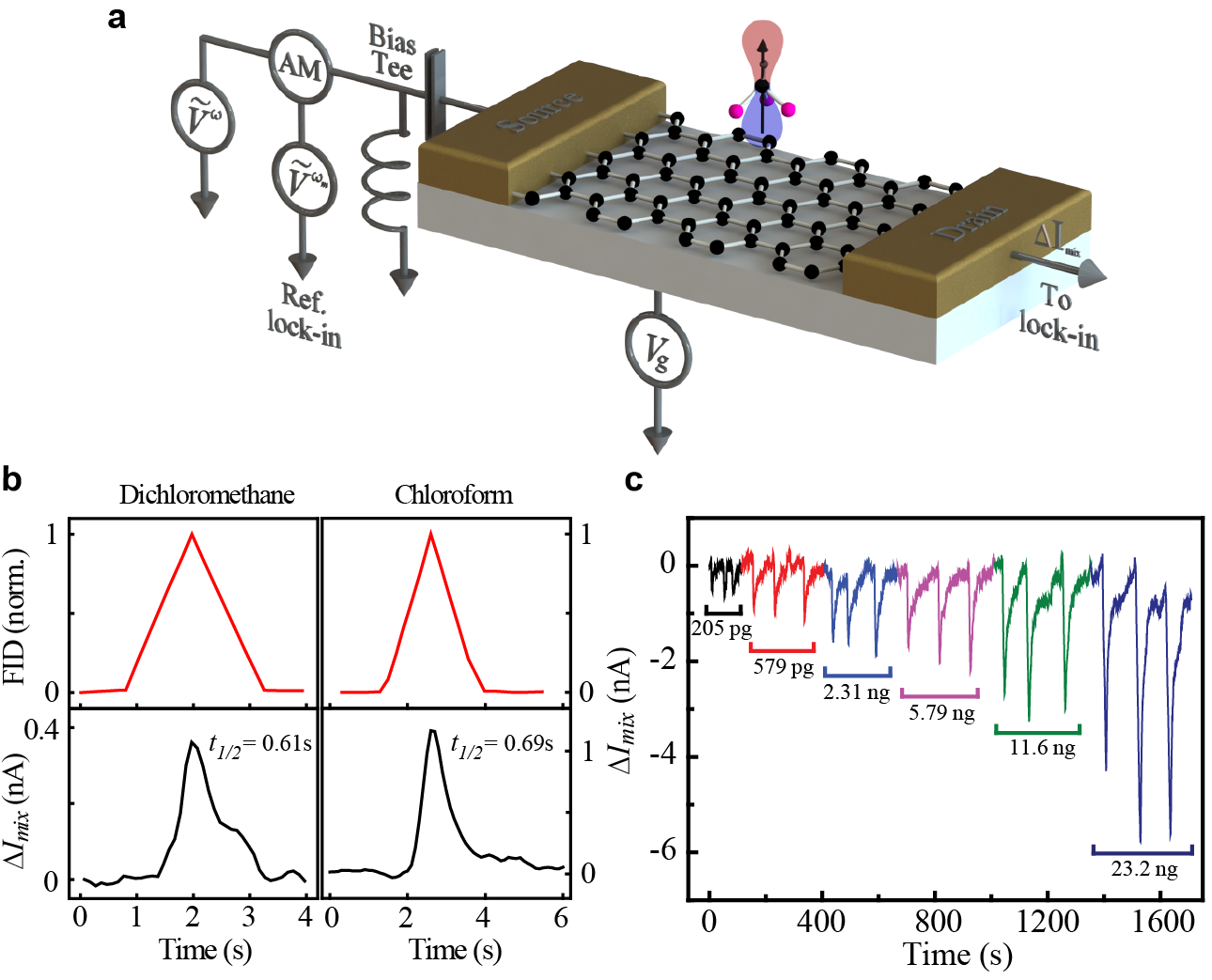
Carbon nanotube high-frequency biosensor
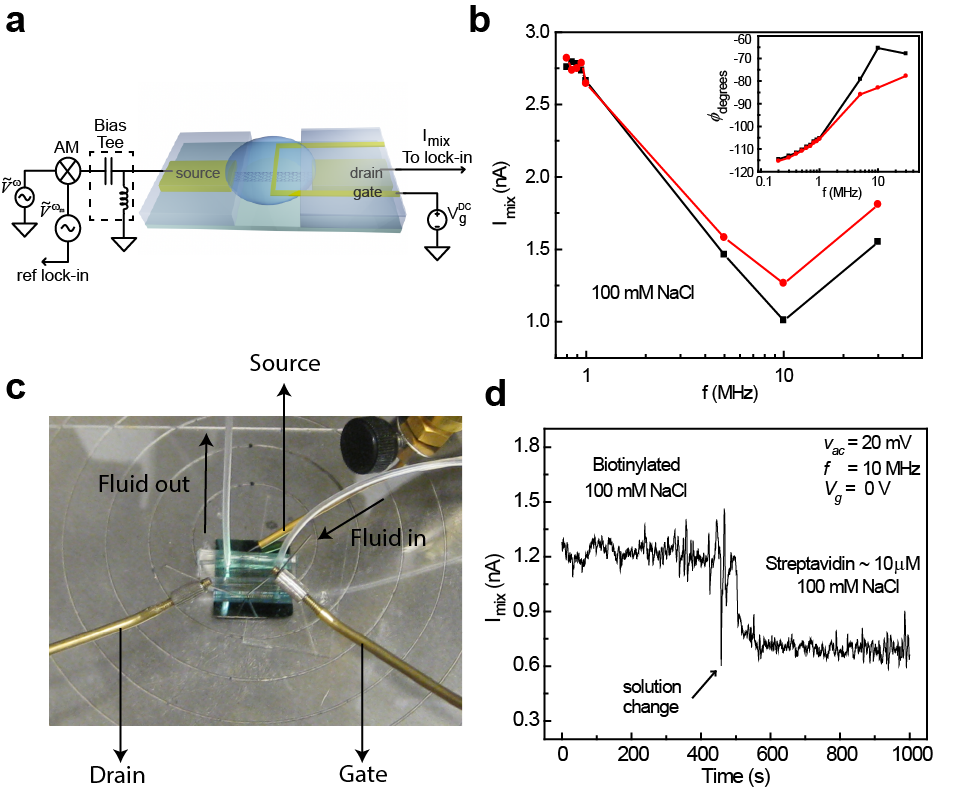
Nanoelectronic platforms are ideal for highly sensitive detection of biological species owing to their large surface-to-volume ratio and unique electronic properties. Consequently, DC (direct current) detection of a wide range of molecules has been successfully demonstrated. However, sensing mechanism based on measuring changes in dc conductance fails at physiologically relevant background salt concentrations, where the sensitivity of the devices suffers from the ionic screening due to mobile ions present in the solution.As the result, most of existing nanoelectronic sensors operate either in dry state or in low ionic strength solutions (c ~ 1 nM to 10 mM). We demonstrate that the fundamental ionic screening effect can be mitigated by operating single-walled carbon nanotube field effect transistors as high-frequency biosensors where the ac driving force can no longer overcome the solution drag and the ions in solution do not have sufficient time to form the electrostatic double layer (EDL). As the EDL is weakened, the AC electric field penetrates deeper into the solution, and can drive the dipoles of target biomolecular species, which in turn act as local oscillating gate.
Kulkarni and Zhong. Nano Letters, 12 (2), pp. 719-723, 2012.Kulkarni and Zhong. J. Vis. Exp. (77), e50438, doi: 10.3791/50438, 2013.