|
Motor proteins are enzymes that
convert chemical energy into mechanical work in the cell. They
perform a multitude of vital roles including motility, cell
division, long-range transport, ATP production, and sensing, and
are present in all forms of life from bacteria to humans. For
instance, the motor protein myosin-1 is the active element in
muscle contraction. Bacterial flagella are powered by rotary motor
proteins. Another type of motor protein, dynein, powers flagella
and cillia in eukaryotes, such as in sperm. The motor protein
prestin is thought to provide the feedback mechanism that
increases frequency differentiation and sensitivity in hearing.
During mitosis, motor proteins are implicated in the positioning
of chromosomes during metaphase. Another motor protein then
cleaves the two halves apart in anaphase. The ubiquity of motor
proteins in biology makes understanding their mechanisms of the
utmost importance.
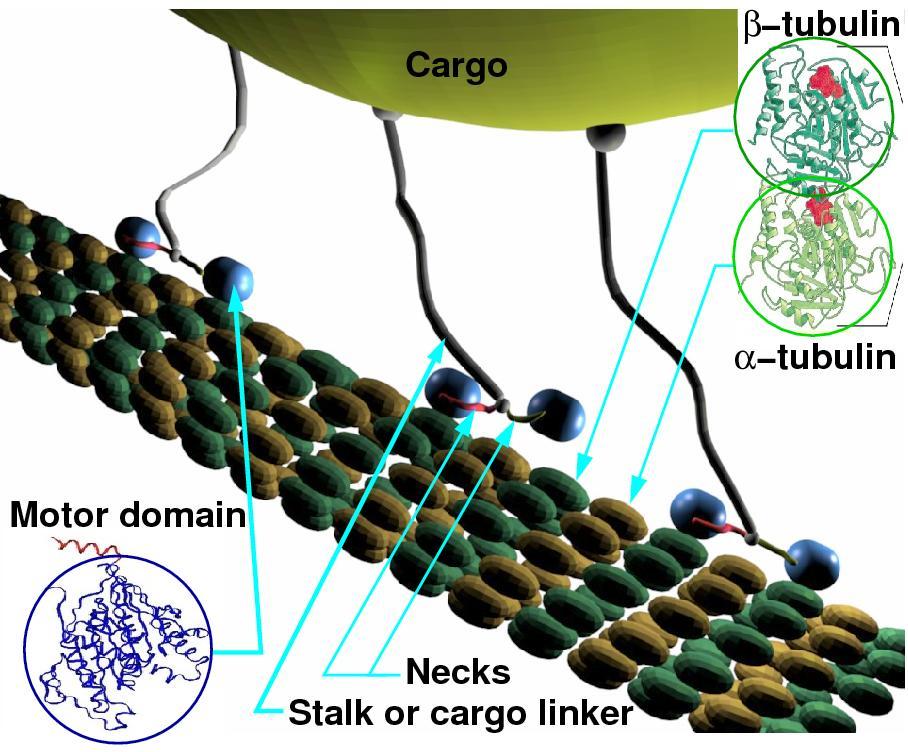
We are currently focused on
kinesin-1, which is a processive molecular motor that converts the
energy from ATP hydrolysis and Brownian motion into directed
movement. Single-molecule techniques allow the experimental
characterization of kinesin in vitro at a range of loads and ATP
concentrations. Mounting evidence suggests that in the cell,
several kinesin motors work collectively to transport a cargo. We
develop models capable of describing the collective behavior of
several coupled motors.
|

